Vesicle-vacuole fusion – A little less of a mystery
Recently, Yvon Jaillais attended me in his tweet of the latest paper of the Raikhel/Hicks lab. I have been wanting to write a review of this paper as it shows some nice research. However, reading the article I also quickly came to realise, that in order to avoid losing you, I needed some visualisation of all the different interactions that take place. So it took me a bit longer than I hoped for to prepare this highlight. As the figures illustrating the different interactions are mine, all mistakes in the interpretation are mine as well. The paper elegantly describe a set of experiments they have done to decipher a bit more of the PVC/vacuole fusion process. Making use of a small synthetic molecule Endosidin 17 (ES17) that disrupts the fusion process, they found how VPS35-RAB7 interaction plays a role in this process.
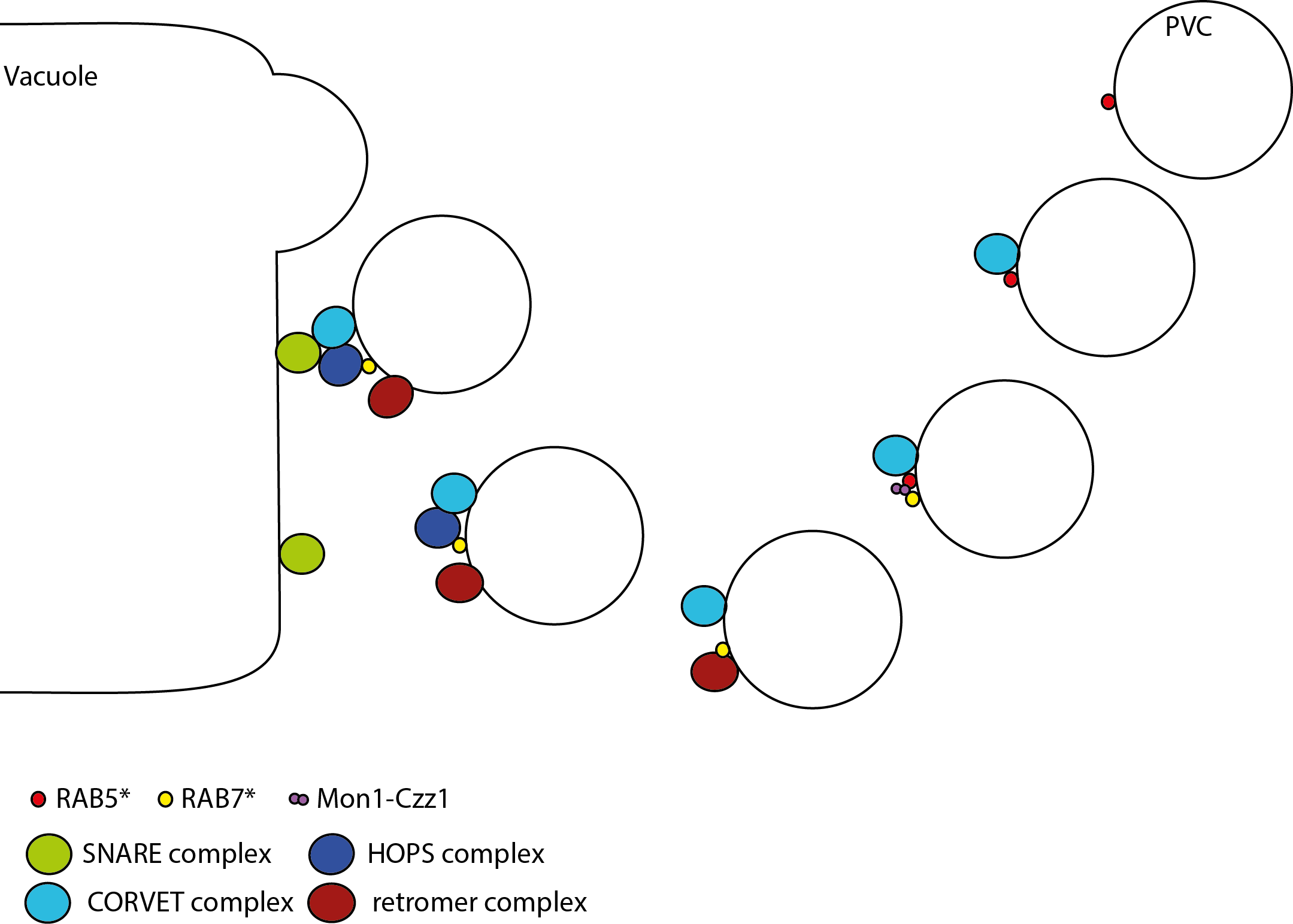
In plant cells vacuoles have big role in keeping the cell working as it does.Vacuoles are not only used as storage compartments for lots of molecules. They also have a role in recycling cellular components like receptors. And can act as compartments were cellular molecules are be broken down. As you can see vacuoles fulfil a diverse set of functions. To avoid of it all ending in chaos trafficking towards the vacuole needs to be tightly regulated. Vesicles are emerging from different parts of the cell, and those that are destined to fuse with the vacuole membrane (tonoplast) are called prevacuolar compartments, PVCs for short. Currently our understanding in how this fusion to happens is by the following steps:
- The RAB-GTPase RAB5 binds to the PVCs and is subsequently activated (RAB5*).
- PVCs-RAB5* recruits the CORVET (VPS8, VPS3, VPS16, VPS18, and VPS33) complex, followed by interaction with VAMP727 and SYP22 of the tonoplast SNARE complex to initiate fusion with the tonoplast.
Additionally
- Recruitment of Mon1-Ccz1 triggers exchange of RAB5 for RAB7 and subsequently activates RAB7 (RAB7*).
- RAB7*-PVC recruits the retromer (VPS35), VPS29, VPS26) complex and interacts with the HOPS (VSP41, VSP39, VPS33, VPS18 and VPS16) tethering complex which subsequent leads to fusion withe the tonoplast, through interaction of VPS33 with SPY22 from the tonoplast SNARE complex
Whereby the exchange of RAB5 for RAB7 and subsequent recruitment of the retromer and HOPS complexes can be seen as one of those additional safety checks to avoid unwanted fusions.
In the recent publication of Raikhel/Hicks lab they investigated this last step in more detail. Using PIN2-GFP as a way to visualise vesicle trafficking they found that ES17 treatment affected PIN2-GFP transport to the vacuole. Using reporter lines for different steps of vesicle transport, they narrowed ES17 effect down to PVCs fusion with the tonoplast.
Following this, they went on to investigate to which protein ES17 was binding and which interaction it disrupted. This led to the finding that ES17 binds to VPS35 at the location where it normally binds RABG3f (a RAB7 protein). The disruption of the interaction between VPSP35 and RABG3f had as result that VPS35 could not anchor itself to the PVC membrane to assemble the retromer complex. Moreover, VPS35-RABG3f interaction was shown to be a requirement for the recruitment of VPS39 and VPS33 of the HOPS complex to the PVC. Which in itself is needed for the fusion of the PVC with the vacuole.
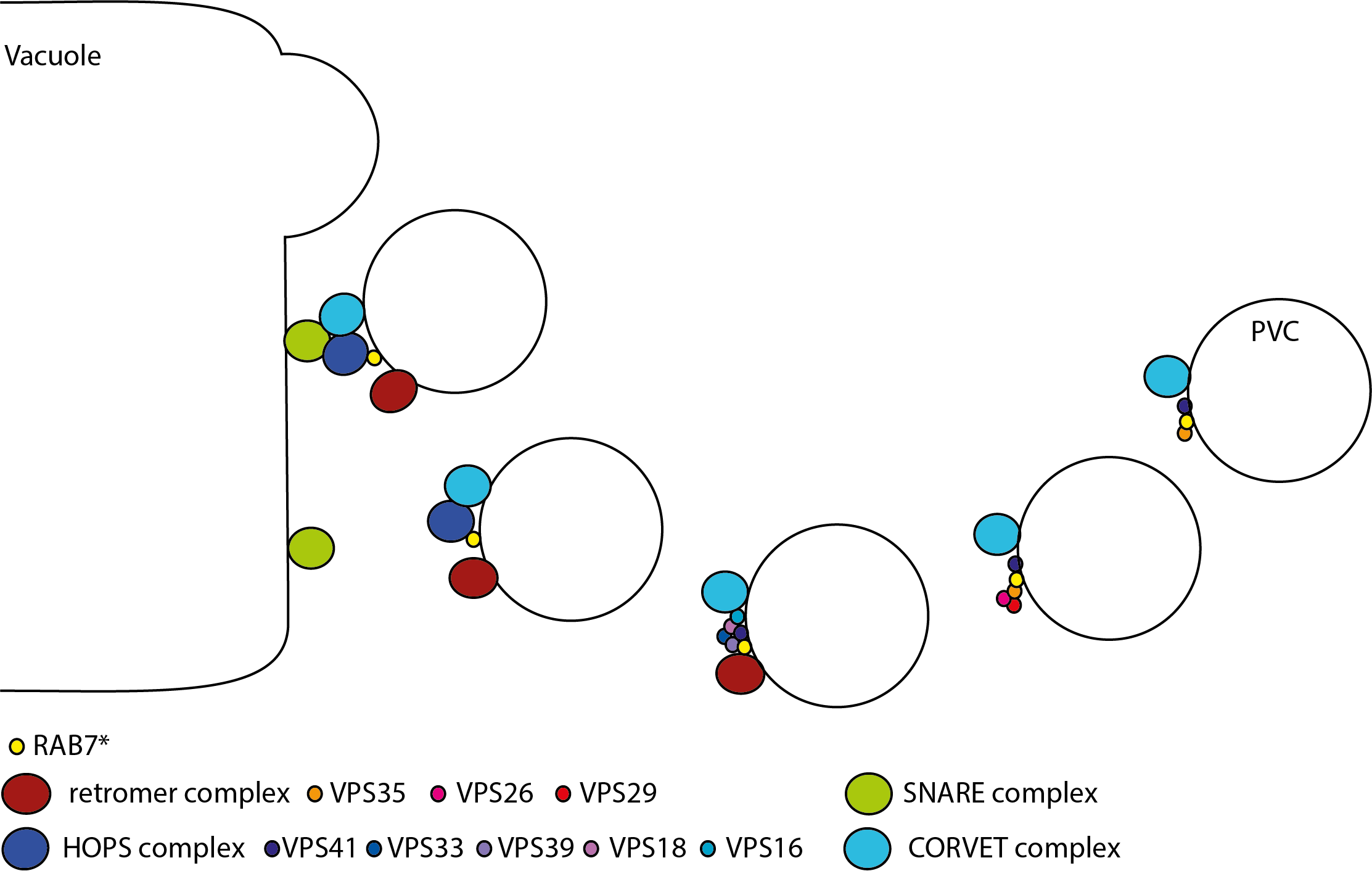
The authors conclude that the interaction of RAB7 with VPS35 and the retromer complex functions as a kind of checkpoint, after which VSP35 presumably releases RRABG3f, so RABG3f becomes available for the recruitment of the HOPS complex and subsequent fusion with the tonoplast.
