SNAREs deal with K+ channels
It has been a busy week so it took some time to get my thoughts organised and write this review. Keeping up with vesicle fusion, we now have a look at how SNARE assembly is regulated.
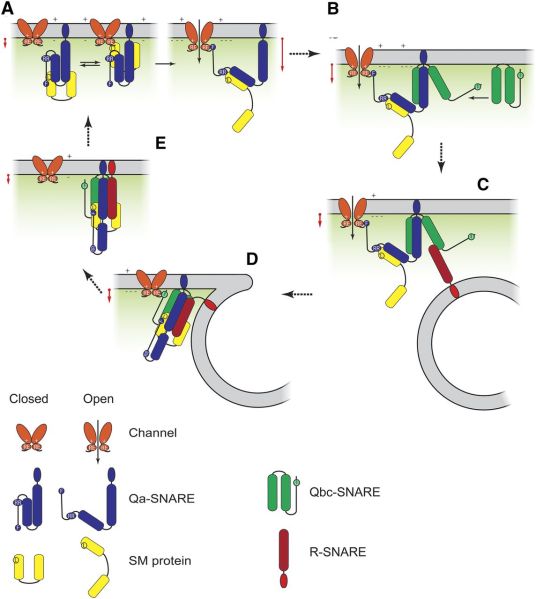
The final step of vesicle fusion with the plasma membrane is regulated by the SNARE complex. This complex is involved in vesicle fusion as follow. Two of the SNARE proteins Qa-SNARE and Qbc-SNARE assemble together with a regulatory SM-protein on the plasma membrane. While the third SNARE protein R-SNARE is located on the vesicle membrane. When the vesicle comes near the plasma membrane the R-SNARE protein is able to interact with the Q-SNARE proteins, bringing the vesicle close to the membrane, enabling fusion. The interaction between Qa-SNARE and Qbc-SNARE is regulated via the open or closed confirmation of Qa-SNARE and its interaction with the SM-protein. However, what causes Qa-SNARE to switch from an closed to open conformation is unclear.
This is were K+ channels come in. Previous research showed that the Arabidopsis Qa-SNARE, SYP121, showed that it binds directly with K+ channels on the plasma membrane, its binding is promoting opening of the channel and K+ uptake. In addition, Arabidopsis R-SNARE, VAMP721, was also shown to bind to K+ channel but instead of promoting K+ uptake it inhibits it. To make matters even more confusing the SPY121 channel binding site overlaps with its SM-protein, SEC11, binding site, which in turn is believed to be important for switching SYP121 from closed to open confirmation.
Now new research from Blatt’s lab is shining new light on the SNARE complex assembly and how its interaction with K+ channels enables coordinating vesicle traffic with K+ uptake. They painstakingly analysed the effects of interaction of SEC11 and each of the SNARE proteins with the K+ channel. This uncovered that all four proteins are able to interact with the K+ channel, and affect the K+ uptake. With SYP121 and VAMP721 strongly competing for K+ channel binding. However, the presence of SEC11 promotes SYP121 binding with the K+ channel, while inhibiting VAMP721 and K+ channel binding. The interaction of the K+ channel with SYP121 brings about a conformational change that turns SYP121 into an open conformation. Which enabled subsequent binding with SNAP33. They subsequently showed that SEC11 stabilises the binding between SYP121 and SNAP33. And that when SNAP33 is present VAMP721 is able to bind the Q-SNARE complex. The assembly of the SNARE complex finishes with the fusion of the vesicle with the membrane.
The authors summarised their findings and proposed mechanism of SNARE assembly with a nice figure, which I would not like to withhold you, as it probably does a better job showing what is going on than my description.
Literature
Waghmare et al., K+ Channel-SEC11 Binding Exchange Regulates SNARE Assembly for Secretory Traffic.
